In a breakthrough for gene therapy, the international academic journal Nature has published a landmark clinical study detailing the successful use of a novel base-editing drug to treat β-thalassemia.
Jointly conducted by ShanghaiTech University, the First Affiliated Hospital of Guangxi Medical University, Fudan University, and CorrectSequence Therapeutics, the early-stage clinical trial of the base-editing drug CS-101 injection reports that all treated transfusion-dependent patients rapidly achieved transfusion independence and regained healthy hematopoietic function.
The first base-editing clinical study ever published in Nature, this milestone firmly establishes China’s original innovation and clinical translation at the global forefront of cutting-edge gene editing therapies.
A pressing unmet medical need
β-thalassemia is a hereditary blood disorder caused by mutations in the β-globin gene, leading to insufficient production of functional hemoglobin. Patients with transfusion-dependent thalassemia (TDT) must undergo blood transfusions every two to five weeks to survive. While life-saving, this treatment carries significant risks—including iron overload and infection—and places a heavy, lifelong burden on patients, families, and healthcare systems.
At present, allogeneic hematopoietic stem cell transplantation is the only curative option. However, its use is limited by donor availability, the risk of immune rejection, and high treatment costs. These challenges highlight the urgent need for safer, more effective therapies.
A new generation of gene editing technology
Gene editing technologies have opened new possibilities for curing monogenic diseases such as β-thalassemia. Unlike conventional CRISPR-Cas9 approaches, often described as “molecular scissors” that cut DNA, the transformer Base Editor (tBE) developed by the team acts more like a “gene correction pen.” It enables precise conversion of specific DNA bases without introducing double-strand breaks.
This approach avoids risks associated with DNA cutting, such as chromosomal deletions or rearrangements, and is expected to offer improved safety.
Rapid and durable clinical responses
The research team evaluated CS-101 in an investigator-initiated trial (IIT) (Figure 1) involving five patients with transfusion-dependent β-thalassemia. As of November 17, 2025, all patients achieved successful neutrophil and platelet engraftment following treatment.
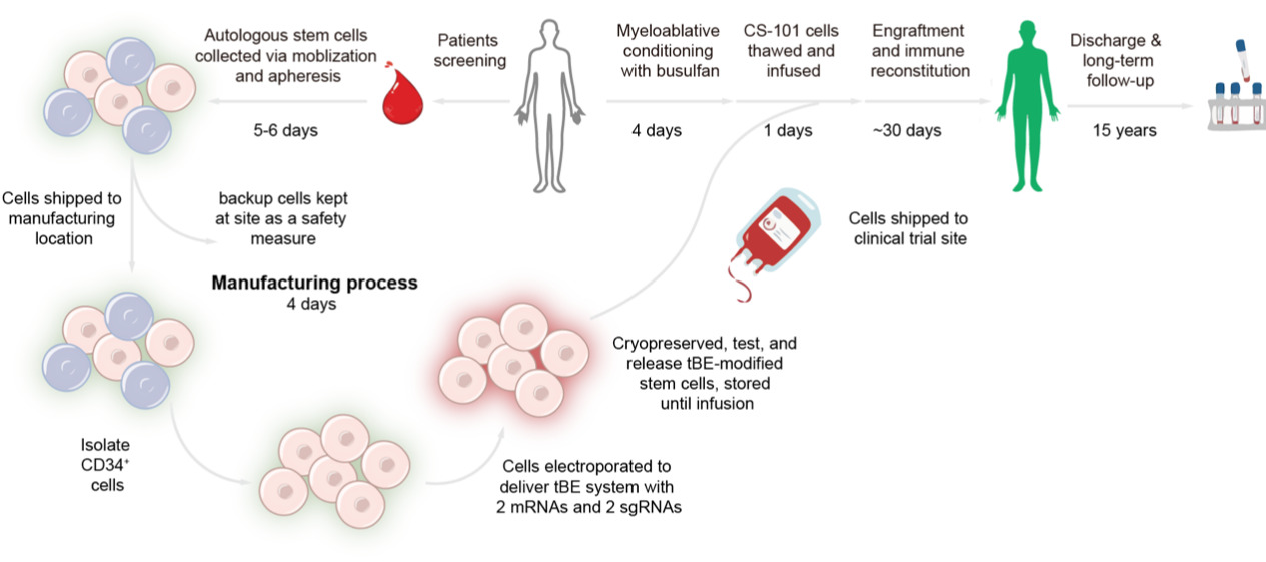
Figure 1: Clinical treatment workflow of CS-101
Remarkably, patients became transfusion-independent within an average of just 16 days after a single dose. Hemoglobin levels rose to near-normal levels (12.4 g/dL) within three months and remained stable at approximately 13.4 g/dL after 15 months of follow-up. All patients have maintained transfusion independence for more than one year, with the longest follow-up exceeding 28 months.
Compared with existing CRISPR-based therapies, CS-101 demonstrated faster activation of fetal hemoglobin (HbF), more rapid hematopoietic recovery, and earlier restoration of normal hemoglobin levels. These advantages translate into shorter hospital stays and reduced healthcare burden. In addition, the therapy achieved higher HbF expression levels and showed no evidence of large genomic deletions, chromosomal abnormalities, or significant off-target effects, indicating a favorable safety profile.
Toward a first-in-class base editing therapy
To date, CS-101 has completed Phase I IND clinical studies and has been used to treat nearly 20 patients with β-thalassemia and sickle cell disease in China and internationally. All treated patients achieved transfusion independence, with sustained hemoglobin expression over time.
CS-101 has the potential to become the world’s first approved base editing therapeutic. With its strong efficacy, safety profile, and cost advantages, this innovative technology holds promise to benefit patients worldwide.

